We asked Yale Medicine infectious diseases experts to answer common questions about these new, reformulated boosters. Studies show that the initial vaccine doses are.  
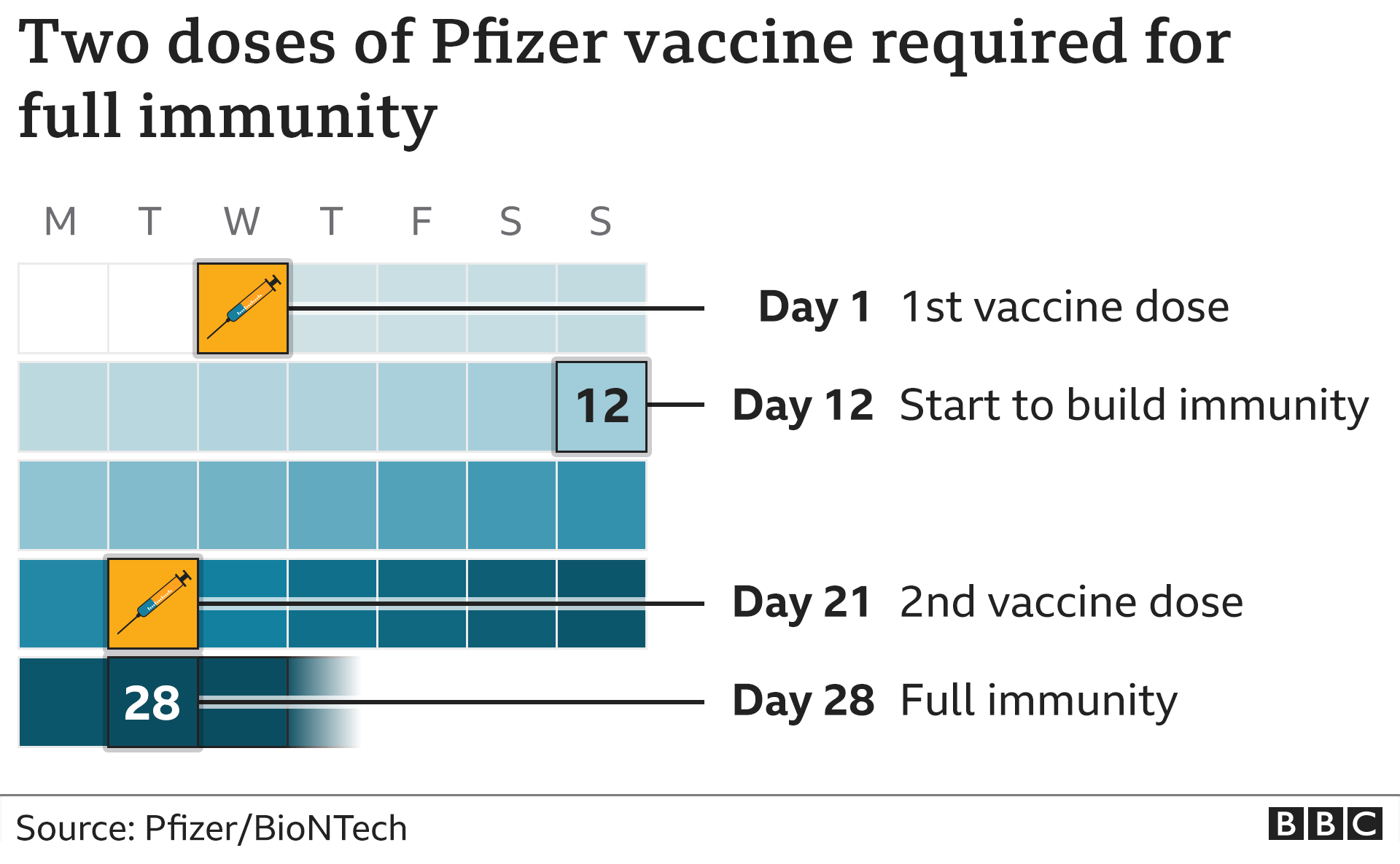
Since the updated boosters (one from Pfizer-BioNTech and the other from Moderna) were granted an FDA emergency use authorization (EUA), the FDA and CDC have approved them for children, starting at age 6 months. Jane Greenhalgh Twitter Enlarge this image Having a compromised immune system puts you at higher risk of severe illness and death from COVID-19. Additional booster doses for 16 to 29 year olds at high risk. The additional dose (s) help your immune system to better protect you against COVID-19 infection. While the original mRNA coronavirus vaccines have proven effective at preventing death and severe disease from COVID-19, breakthrough infections and reinfections have become more common in the face of an evolving virus. its recommended you wait at least 6 months since your last COVID-19 vaccine or infection. People who are aged 6 months and older who are moderately or severely immunocompromised may get 1 additional updated COVID-19 vaccine dose 2 or more months after the last recommended updated COVID-19 vaccine. Children 6 months through 5 years of age who are unvaccinated may receive a two-dose series of the Moderna bivalent vaccine (6 months through 5 years of age) OR. The boosters target two Omicron subvariants, BA.4 and BA.5. In the fall of 2022, the Food and Drug Administration (FDA) authorized-and the Centers for Disease Control and Prevention (CDC) recommended-two updated boosters. When the mRNA COVID-19 vaccines were first unveiled in December 2020, medical experts touted the benefits of this new technology, saying formulations could easily be tweaked someday to match a quickly changing virus. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
